Which of the following compounds can be correctly described as an Arrhenius base when dissolved in water?
- Sodium nitrate
- Sodium sulfate
- Sodium chloride
- Sodium hydroxide
Aussie Maths & Science Teachers: Save your time with SmarterEd
Which of the following compounds can be correctly described as an Arrhenius base when dissolved in water?
\(D\)
\(\Rightarrow D\)
Explain how the following substances would be classified under the Arrhenius and Brønsted-Lowry definitions of acids. Support your answer with relevant equations. (4 marks)
--- 12 WORK AREA LINES (style=lined) ---
Which of the following is NOT a Bronsted-Lowry reaction?
`B`
`=> B`
Which of the following would NOT have been classified as an acid by Antoine Lavoisier in 1780?
`D`
`=>D`
The equipment shown is set up. After some time a ring of white powder is seen to form on the inside of the glass tube.
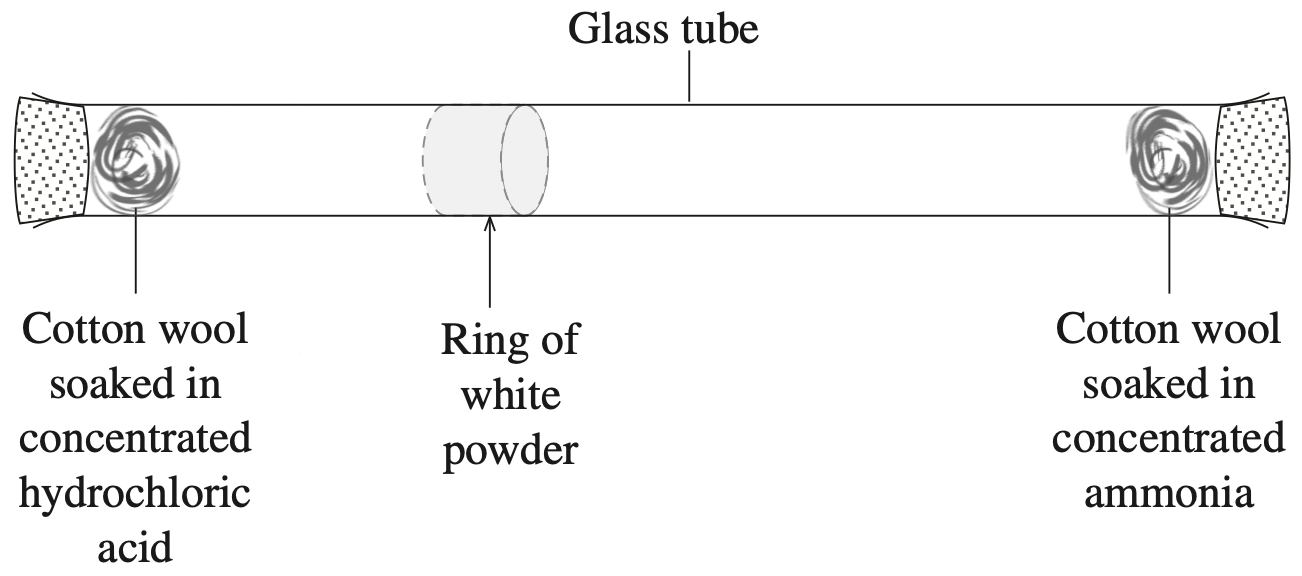
--- 4 WORK AREA LINES (style=lined) ---
--- 4 WORK AREA LINES (style=lined) ---
a. According to Arrhenius:
b. \(\ce{HCl(g) + NH3(g) -> NH4+ + Cl-}\)
a. According to Arrhenius:
b. \(\ce{HCl(g) + NH3(g) -> NH4+ + Cl-}\)
Assess the usefulness of the Brønsted-Lowry model in classifying acids and bases. Support your answer with at least TWO chemical equations. (5 marks)
Which of the following is an Arrhenius base?
`B`
`=>B`
The effect of concentration on the pH of acrylic acid `(text{C}_2 text{H}_3 text{COOH})` and hydrochloric acid `(text{HCl})` solutions is shown in the graph. Both of these acids are monoprotic.
Explain the trends in the graph. Include relevant chemical equations in your answer. (4 marks)
\(\ce{HCl(aq) -> H+ (aq) + Cl– (aq)}\)
\(\ce{C2H3COOH(aq) \rightleftharpoons H+ (aq) + C2H3COO– (aq)}\)
\(\ce{HCl(aq) -> H+ (aq) + Cl– (aq)}\)
\(\ce{C2H3COOH(aq) \rightleftharpoons H+ (aq) + C2H3COO– (aq)}\)
The pH of two aqueous solutions was compared.
Explain why the `\text{HCN}(aq)` solution has a higher pH than the `\text{HCl}(aq)` solution. Include a relevant chemical equation for the `\text{HCN}(aq)` solution. (3 marks)
--- 9 WORK AREA LINES (style=lined) ---
The following equation describes an equilibrium reaction.
\( \ce{HF(aq) + PO4^3-(aq) \rightleftharpoons HPO4^2-(aq) + F-(aq)} \)
Identify ONE base and its conjugate acid in the above equation. (2 marks)
--- 0 WORK AREA LINES (style=lined) ---
Possible answers:
\begin{array} {ll}
\text{Base} & \text{Conjugate Acid} \\
\hline \ce{PO4^3-(aq)} & \ce{HPO4^2-(aq)} \\
\ce{F-(aq)} & \ce{HF(aq)} \\
\end{array}
Possible answers:
\begin{array} {ll}
\text{Base} & \text{Conjugate Acid} \\
\hline \ce{PO4^3-(aq)} & \ce{HPO4^2-(aq)} \\
\ce{F-(aq)} & \ce{HF(aq)} \\
\end{array}