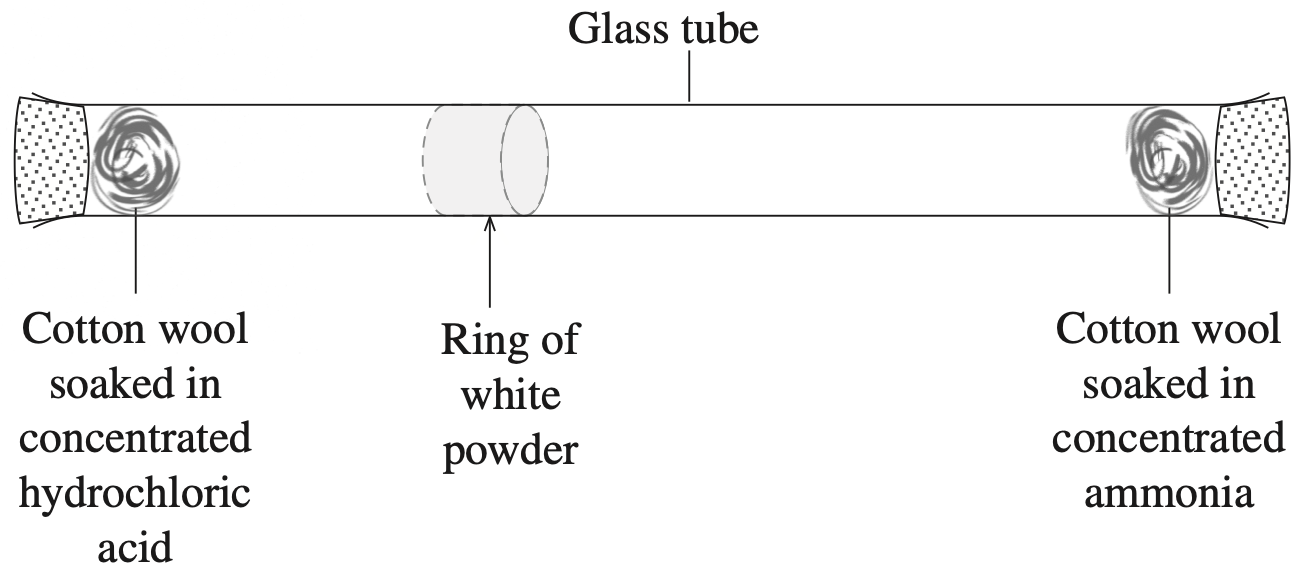
Explain how the following substances would be classified under the Arrhenius and Brønsted-Lowry definitions of acids. Support your answer with relevant equations. (4 marks)
-
- \( \ce{HCl(aq)} \)
- \( \ce{NH4Cl(aq)} \)
--- 12 WORK AREA LINES (style=lined) ---
→ Acids are defined by Arrhenius as hydrogen-containing compounds that dissociate in water to give \(\ce{H+}\) ions.
→ \(\ce{HCl(aq)}\) produces \(\ce{H+}\) ions in water and therefore qualifies within Arrhenius’ definition of an acid.
\(\ce{HCl(aq) \rightarrow H+(aq) + Cl−(aq)}\)
→ The salt \(\ce{NH4Cl}\) would not be recognised as an acid with Arrhenius’ definition, since the predominant ions present in aqueous solution are ammonium and chloride.
→ The Brønsted−Lowry theory states that acids are proton donors. \(\ce{HCl(aq)}\) is a proton donor and therefore also qualifies as a Brønsted−Lowry acid.
→ In contradiction to Arrhenius, ammonium chloride \(\ce{(NH4Cl)}\) is also classified as a Brønsted−Lowry acid. This is due to the ammonium ion donating a proton to water to form a hydronium ion.
\(\ce{NH4+(aq) + H2O(l) \rightleftharpoons NH3(aq) + H3O+(aq)}\)
→ Acids are defined by Arrhenius as hydrogen-containing compounds that dissociate in water to give \(\ce{H+}\) ions.
→ \(\ce{HCl(aq)}\) produces \(\ce{H+}\) ions in water and therefore qualifies within Arrhenius’ definition of an acid.
\(\ce{HCl(aq) \rightarrow H+(aq) + Cl−(aq)}\)
→ The salt \(\ce{NH4Cl}\) would not be recognised as an acid with Arrhenius’ definition, since the predominant ions present in aqueous solution are ammonium and chloride.
→ The Brønsted−Lowry theory states that acids are proton donors. \(\ce{HCl(aq)}\) is a proton donor and therefore also qualifies as a Brønsted−Lowry acid.
→ In contradiction to Arrhenius, ammonium chloride \(\ce{(NH4Cl)}\) is also classified as a Brønsted−Lowry acid. This is due to the ammonium ion donating a proton to water to form a hydronium ion.
\(\ce{NH4+(aq) + H2O(l) \rightleftharpoons NH3(aq) + H3O+(aq)}\)