The molar heat of combustion of pentan-1-ol is 2800 kJ mol−1. A quantity of pentan-1-ol was combusted, generating 108 kJ of heat. \(MM=88.146\ \text{gmol}^{-1}\)
What mass of pentan-1-ol was combusted? (2 marks)
Aussie Maths & Science Teachers: Save your time with SmarterEd
The molar heat of combustion of pentan-1-ol is 2800 kJ mol−1. A quantity of pentan-1-ol was combusted, generating 108 kJ of heat. \(MM=88.146\ \text{gmol}^{-1}\)
What mass of pentan-1-ol was combusted? (2 marks)
3.40 grams
\(\ce{Pentan-1-ol\ \Rightarrow C5H12O}\)
\(\ce{MM(C5H12O) = 12.01 \times 5 + 1.008 \times 12 + 16.00 = 88.146\ \text{g mol}^{-1}}\)
\(\ce{Moles combusted = \dfrac{108}{2800} = 0.03857\ \text{mol}}\)
\(\ce{m(C5H12O) = 0.03857 \times 88.146 = 3.40\ \text{g}}\)
The table shows the heat of combustion for four compounds.
| \(\textit{Compound}\) | \(\textit{Heat of combustion}\) \( (\text{kJ mol}^{-1} )\) |
| \(\ce{CO}\) | \(233\) |
| \(\ce{CH4}\) | \(890\) |
| \(\ce{C2H2}\) | \(1300\) |
| \(\ce{C2H6}\) | \(1560\) |
Which of these compounds would produce the greatest amount of energy if 1.00 g of each is burnt?
\(B\)
→ The smallest molecular mass it will result in the highest number of moles.
→ The highest number of moles gives the greatest amount of energy.
→ By comparing the molecular mass of each compound, the smallest is methane (\(\ce{CH4})\).
\(\Rightarrow B\)
The heat of combustion of propan-1-ol is 2021 kJ mol−1. Combustion takes place according to the equation:
\(\ce{2C3H7OH(l) + 9O2(g) \rightarrow 6CO2(g) + 8H2O(l)}\)
What mass of water is formed when 1530 kJ of energy is released? (2 marks)
--- 5 WORK AREA LINES (style=lined) ---
\(54.55\ \text{g}\)
→ The stoichiometric ratio between propanol and water = 1 : 4
→ Moles of propane required if 1530 kJ of energy released:
\(\ce{n(C3H7OH) = \dfrac{1530}{2021} = 0.757\ \text{mol}}\)
\(\ce{n(H2O) = 4 \times 0.757 = 3.028\ \text{mol}}\)
\(\ce{MM(H2O) = 2 \times 1.008 + 16 = 18.016\ \text{g mol}^{-1}}\)
\(\ce{m(H2O) = 3.028 \times 18.016 = 54.55\ \text{g (2 d.p.)}}\)
Hydrogen peroxide, \(\ce{H2O2}\), in aqueous solution at room temperature decomposes slowly and irreversibly to form water, \(\ce{H2O}\), and oxygen, \(\ce{O2}\), according to the following equation. \(\ce{2H2O2(aq)\rightarrow 2H2O(l) + O2(g)}\) \(\Delta H < 0\) --- 6 WORK AREA LINES (style=lined) --- --- 1 WORK AREA LINES (style=lined) --- a. → Increasing the temperature will increase the rate of \(\ce{O2}\) production. → An increase in temperature increases the kinetic energy of all molecules which causes both the frequency of collisions and the energy of each collision to increase. → The temperature increase results in more collisions, of which a higher proportion are successful (i.e. occur with greater energy than the activation energy threshold required to react). b. \(\ce{MnO2}\) acts as a catalyst in this reaction. a. → Increasing the temperature will increase the rate of \(\ce{O2}\) production. → An increase in temperature increases the kinetic energy of all molecules which causes both the frequency of collisions and the energy of each collision to increase. → The temperature increase results in more collisions, of which a higher proportion are successful (i.e. occur with greater energy than the activation energy threshold required to react). b. \(\ce{MnO2}\) acts as a catalyst in this reaction.
A coal-fired power station is used to generate electricity. Carbon dioxide, \(\ce{CO2}\), gas is produced as part of the process.
Carbon capture and storage is one option being considered to reduce the amount of \(\ce{CO2}\) released into the atmosphere. 5.17 × 10\(^4\) mol of \(\ce{CO2}\) is captured, cooled to 28 °C and stored in a sealed 20 000.0 L tank prior to transportation.
Calculate the pressure, in kilopascals, in the tank when it contains 5.17 × 10\(^4\) mol of \(\ce{CO2}\). (2 marks)
\(6.47 \times 10^{3}\ \text{kPa}\)
\(\ce{n(CO2) = 5.17 \times 10^{4} \ \text{mol (given)}}\)
\(\ce{T = 273 + 28 = 301\ \text{K}}\)
\(\ce{\text{Pressure}\ = \dfrac{\text{nRT}}{\text{V}} = \dfrac{5.17 \times 10^{4} \times 8.31 \times 301}{20 \times 10^{3}} = 6.47 \times 10^{3}\ \text{kPa}}\)
Butane, \(\ce{C4H10}\), undergoes complete combustion according to the following equation.
\(\ce{2C4H10(g) + 13O2(g)\rightarrow 8CO2(g) + 10H2O(g)}\)
67.0 g of \(\ce{C4H10}\) released 3330 kJ of energy during complete combustion at standard laboratory conditions (SLC).
Calculate the mass of carbon dioxide, \(\ce{CO2}\), produced in this reaction. (2 marks)
\(202.8\ \text{g}\)
\(\ce{MM(C4H10) = 4 \times 12.01 + 10 \times 1.008 = 58.1\ \text{g mol}^{-1}}\)
\(\ce{n(C4H10) = \dfrac{67.0}{58.1} = 1.153\ \text{mol}}\)
\(\ce{n(CO2) = 4 \times 1.153 = 4.61\ \text{mol}}\)
\(\ce{MM(CO2) = 12.01 + 2 \times 16 = 44.0\ \text{g mol}^{-1}}\)
\(\ce{m(CO2) = 4.61 \times 44.0 = 202.8\ \text{g}}\)
If 20.0 g of methane gas, \(\ce{CH4}\), is kept in a 5.0 L sealed container at 25 °C, what would be the pressure in the container? (2 marks)
\(6.2 \times 10^{2}\ \text{kPa}\)
\(\ce{MM(CH4) = 12.01 + 4 \times 1.008 = 16.0\ \text{g mol}^{-1}}\)
\(\ce{n(CH4) = \dfrac{\ce{m}}{\ce{MM}}= \dfrac{20.0}{16.0} = 1.25\ \text{mol}}\)
\(\ce{\text{Pressure} (CH4) = \dfrac{\text{nRT}}{\text{V}} = \dfrac{1.25 \times 8.31 \times 298}{5.0} = 6.2 \times 10^{2}\ \text{kPa}}\)
Standard solutions of sodium hydroxide, \(\ce{NaOH}\), must be kept in airtight containers. This is because \(\ce{NaOH}\) is a strong base and absorbs acidic oxides, such as carbon dioxide, \(\ce{CO2}\), from the air and reacts with them. As a result, the concentration of \(\ce{NaOH}\) is changed to an unknown extent.
\(\ce{CO2}\) in the air reacts with water to form carbonic acid, \(\ce{H2CO3}\). This can react with \(\ce{NaOH}\) to form sodium carbonate, \(\ce{Na2CO3}\).
--- 1 WORK AREA LINES (style=lined) ---
--- 1 WORK AREA LINES (style=lined) ---
i. \(\ce{CO2(g) + H2O(l) \rightarrow H2CO3(aq)} \)
ii. \(\ce{2NaOH(aq) + H2CO3(aq) \rightarrow Na2CO3(aq) + 2H2O(l)}\)
i. \(\ce{CO2(g) + H2O(l) \rightarrow H2CO3(aq)} \)
ii. \(\ce{2NaOH(aq) + H2CO3(aq) \rightarrow Na2CO3(aq) + 2H2O(l)}\)
A condensation reaction involving 200 glucose molecules, \(\ce{C6H12O6}\), results in a polysaccharide. The molar mass, in g mol\(^{-1}\), of the polysaccharide is
\(C\)
→ 200 \(\ce{C6H12O6}\) molecules react to form a polysaccharide
→ 199 \(\ce{H2O}\) molecules are released in this condensation polymerisation
\(\ce{MM(C6H12O6) = 180.0\ \text{g mol}^{-1} }\)
\(\ce{MM(H2O) = 18.0\ \text{g mol}^{-1} }\)
| \(\ce{MM(\text{polysaccharide})}\) | \(= 200 \times 180.0-199 \times 18.0\) | |
| \(= 32\ 418\ \text{g mol}^{-1}\) |
\(\Rightarrow C\)
This set of data was obtained from a motion investigation to determine the acceleration due to gravity on a planet other than Earth. --- 5 WORK AREA LINES (style=lined) ---
Time (s)
Vertical velocity (m s\(^{-1}\))
0.60
0.02
1.00
0.09
1.20
0.12
1.40
0.17
1.80
0.23
Plot the data from the table, and then calculate the acceleration. (3 marks)
Industrially, ethanol, \(\ce{C2H5OH} \), is made by either of two methods.
One method uses ethene, \(\ce{C2H4} \), which is derived from crude oil.
The other method uses a sugar, such as sucrose, \(\ce{C12H22O11}\), and yeast, in aqueous solution.
The production of \(\ce{C2H5OH}\) from \(\ce{C12H22O11}\) and yeast proceeds according to the equation
\( \ce{C12H22O11(aq) + H2O(l) \rightarrow 4C2H5OH(aq) + 4CO2(g)} \)
\(\ce{MM(C12H22O11) = 342\ \text{g mol}^{-1} }\) (2 marks)
--- 4 WORK AREA LINES (style=lined) ---
\(\ce{C2H4(g) + \text{insert box} ->[\text{catalyst}] C2H5OH(g) }\) (1 mark)
--- 5 WORK AREA LINES (style=lined) ---
--- 5 WORK AREA LINES (style=lined) ---
a. \(\ce{n(C12H22O11) = \dfrac{1250}{342} = 3.65\ \text{mol}} \)
\(\ce{n(C2H5OH) = 4 \times 3.65 = 14.6\ \text{mol}} \)
\(\ce{MM(C2H5OH) = 2 \times 12.01 + 6 \times 1.008 + 16 = 46.07}\)
\(\ce{m(C2H5OH) = 14.6 \times 46.07 = 673\ \text{g (3 sig fig)}} \)
b.i. \(\ce{C2H4(g) + H2O(g) ->[\text{catalyst}] C2H5OH(g) }\)
b.ii. Addition reaction.
Outline a first-hand investigation to demonstrate the transfer of light by optical fibres (2 marks) --- 6 WORK AREA LINES (style=lined) --- → Using a laser beam or other light source, shine the light into a glass rod at an angle at an angle greater than the critical angle. → The light beam can then be seen to reflect off either side of the glass rod as it travels down the line of the rod. → This is called total internal reflection (TIR) and demonstrates effectively how light travels through a optical fibre, As seen in the diagram below. → Using a laser beam or other light source, shine the light into a glass rod at an angle at an angle greater than the critical angle. → The light beam can then be seen to reflect off either side of the glass rod as it travels down the line of the rod. → This is called total internal reflection (TIR) and demonstrates effectively how light travels through a optical fibre, As seen in the diagram below.
An electric field is produced between two charged parallel plates, \(M\) and \(N\).
The plates, \(M\) and \(N\), are 1.0 cm apart and have an electric field of 15 V m\(^{-1}\).
Calculate the potential difference between the plates. (2 marks)
--- 4 WORK AREA LINES (style=lined) ---
\(0.15 \text{ V}\)
\(\text{Convert units: 1 cm = 0.01 m}\).
| \(E\) | \(=\dfrac{V}{d}\) | |
| \(V\) | \(=Ed\) | |
| \(=15 \times 0.01\) | ||
| \(=0.15 \text{ V}\) |
The table gives the heat of combustion of three different alcohols at 25°C.
| \( Alcohol \) | \( Heat \ of \ combustion \) \( \text{(Kj g} ^{-1}) \) |
| \( \text{Methanol} \) | \(22.68\) |
| \( \text{Ethanol} \) | \(29.67\) |
| \( \text{Butan-1-ol} \) | \(36.11\) |
\(B\)
→ The boiling point of straight-chained alcohols increases with their chain length.
→ Therefore the boiling point of Propan-1-ol will exist between Ethanol and Butan-1-ol
\(\Rightarrow B\)
The table shows four separate tests used to identify a dilute, aqueous sample of a compound.
\begin{array} {|c|l|l|}
\hline
\rule{0pt}{2.5ex} \textbf{Test Number} \rule[-1ex]{0pt}{0pt} & \quad \quad \quad \quad \quad \textbf{Test} & \quad \quad \quad \textbf{Observation}\\
\hline
\rule{0pt}{2.5ex} 1\rule[-1ex]{0pt}{0pt} & \text{Test with Red litmus} & \text{Stays red}\\
\hline
\rule{0pt}{2.5ex} 2 \rule[-1ex]{0pt}{0pt} & \text{Add \( \ce{Ba}^{2+} \) ions to a sample} & \text{White precipitate formed}\\
\hline
\rule{0pt}{2.5ex} 3 \rule[-1ex]{0pt}{0pt} & \text{Add \( \ce{OH}^{-} \) ions to a sample}& \text{Brown precipitate formed} \\
\hline
\rule{0pt}{2.5ex} 4 \rule[-1ex]{0pt}{0pt} & \text{Add \( \ce{Cl}^{-}\) ions to a sample}& \text{White precipitate formed}\\
\hline
\end{array}
Which compound would produce the observations shown?
\(A\)
\(\Rightarrow A\)
An indicator solution was obtained by boiling a flower in water.

Two solutions were tested with this indicator.
Which row of the table correctly identifies the colour of each solution?
| \( \ce{H2SO4} \ (1 \ × \ 10^{-5} \ \text{mol L}^{-1}) \) | \( \ce{NaOH} \ (5 \ × \ 10^{-5} \ \text{mol L}^{-1}) \) | |
| \(\text{A.}\) | \( \text{Red} \) | \( \text{Green-yellow} \) |
| \(\text{B.}\) | \( \text{Red} \) | \( \text{Blue-green} \) |
| \(\text{C.}\) | \( \text{Purple} \) | \( \text{Blue-green} \) |
| \(\text{D.}\) | \( \text{Purple} \) | \( \text{Green-yellow} \) |
\(C\)
→ pH can be calculated from the \(\ce{H2SO4}\) and \(\ce{NaOH}\) concentrations, allowing the colour of the solutions to be determined
\(\Rightarrow C\)
Which of the following correctly lists the compounds in order of increasing boiling point?
\(A\)
→ Compounds with functional groups capable of hydrogen bonding have higher boiling points (due to stronger bonds, more energy is required to break)
\(\Rightarrow A\)
A titration was performed using two solutions of equal concentration, producing the following titration curve.
Which combination of solutions does the titration curve represent?
\(C\)
→ Weak base acts as a buffer, resisting an immediate decrease in pH upon addition of strong acid
\(\Rightarrow C\)
A mixture of 0.8 mol of \( \ce{CO} \text{(g)} \) and 0.8 mol of \( \ce{H2} \text{(g)} \) was placed in a sealed 1.0 L container. The following reaction occurred.
\( \ce{CO} \text{(g)} + 2 \ce{H2} \text{(g)} \rightleftharpoons \ce{CH3} \ce{OH}\text{(g)} \)
When equilibrium was established, the mixture contained 0.5 mol of \( \ce{CO} \text{(g)} \).
What amount of \( \ce{H2} \text{(g)} \) was present at equilibrium?
\(A\)
\( \ce{CO} \text{(g)} + 2 \ce{H2} \text{(g)} \rightleftharpoons \ce{CH3} \ce{OH}\text{(g)} \)
\begin{array} {|l|c|c|c|}
\hline & \ce{CO(g)} & \ce{2H2(g)} & \ce{CH3OH} \\
\hline \text{Initial} & 0.8 & 0.8 & 0 \\
\hline \text{Change} & -x & -2x & +x \\
\hline \text{Equilibrium} & 0.5 & 0.2 & 0.03 \\
\hline \end{array}
\(x = 0.3\)
\(\Rightarrow A\)
Two charged plates are initially separated by a distance as shown in the diagram.
The potential difference between the plates remains constant.
Which of the graphs best represents the change in electric field strength as the distance between the two plates is increased?
→The electric field strength and distance vary according to the equation:
\(E=\dfrac{V}{d}\)
→ Thus \(E\) and \(d\) are inversely proportionally which is depicted in \(B\).
\(\Rightarrow B\)
The diagram shows a saucepan of water on an induction cooktop.
Which row of the table correctly identifies a property of the material used to make the saucepan and the frequency of the changing magnetic field produced by the coil?
| Property of saucepan | Frequency | |
| A. | Insulator | High (50 kHz) |
| B. | Conductor | High (50 kHz) |
| C. | Insulator | Low (50 Hz) |
| D. | Conductor | Low (50 Hz) |
\(B\)
→ The saucepan needs to be able to transfer heat from the induction cooktop to the water. The heat is generated from the production of electrical currents in the saucepan, hence it needs to be a conductor.
→ The greater the frequency of the changing magnetic field in the coil correlates to a greater electrical current in the saucepan, hence it will heat up more quickly.
\(\Rightarrow B\)
An organic reaction pathway involving compounds \(\text{A, B,}\) and \(\text{C}\) is shown in the flow chart.
The molar mass of \(\text{A}\) is 84.156 g mol\(^{-1}\).
A chemist obtained some spectral data for the compounds as shown.
| \( \text{Data from} \ ^{1} \text{H NMR spectrum of compound C} \) | ||
| \( Chemical \ Shift \ \text{(ppm)} \) | \( Relative \ peak \ area \) | \( Splitting \ pattern \) |
| \(1.01\) | \(3\) | \(\text{Triplet}\) |
| \(1.05\) | \(3\) | \(\text{Triplet}\) |
| \(1.65\) | \(2\) | \(\text{Multiplet}\) |
| \(2.42\) | \(2\) | \(\text{Triplet}\) |
| \(2.46\) | \(2\) | \(\text{Quartet}\) |
| \( ^{1} \text{H NMR chemical shift data}\) | |
| \( Type \ of \ proton \) | \( \text{δ/ppm} \) |
| \( \ce{R - C\textbf{H}3,R - C\textbf{H}2 - R}\) | \(0.7-1.7\) |
| \( \left.\begin{array}{l}\ce{\textbf{H}3C - CO - \\-C\textbf{H}2 - CO -}\end{array}\right\} \begin{aligned} & \text { (aldehydes, ketones,} \\ &\text{carboxylic acids or esters) }\end{aligned}\) | \(2.0-2.6\) |
| \( \ce{R - C\textbf{H}O} \) | \(9.4-10.00\) |
| \( \ce{R - COO\textbf{H}} \) | \(9.0-13.0\) |
Identify the functional group present in each of compounds \(\text{A}\) to \(\text{C}\) and draw the structure of each compound. Justify your answer with reference to the information provided. (9 marks)
--- 28 WORK AREA LINES (style=lined) ---
Compound \(\text{A}\): Alkene
Compound \(\text{B}\): Secondary alcohol
Compound \(\text{C}\): Ketone
Reasoning as follows:
→ Compound \(\text{A}\) is able to undergo an addition reaction to add water across a \(\ce{C=C}\) bond \(\Rightarrow \) Alkene
→ Compound \(\text{B}\) is the product of the above hydration reaction and is therefore an alcohol.
→ The \(\ce{^{13}C\ NMR}\) spectrum of Compound \(\text{A}\) confirms it is an alkene (132 ppm peak corresponding to the \(\ce{C=C}\) atoms). 3 spectrum peaks indicate 3 carbon environments. The molar mass of compound \(\text{A}\) is 84.156 g mol\(^{-1}\) which suggests symmetry within the molecule.
→ The Infrared Spectrum of Compound \(\text{B}\) has a broad peak at approximately 3400 cm\(^{-1}\). This indicates the presence of an hydroxyl group and confirms \(\text{B}\) is an alcohol.
→ Compound \(\text{C}\) is produced by the oxidation of Compound \(\text{B}\) with acidified potassium permanganate.
→ Compound \(\text{C}\) is a carboxylic acid if \(\text{B}\) is a primary alcohol or a ketone if \(\text{B}\) is a secondary alcohol.
→ Since the \(\ce{^{1}H NMR}\) spectrum of \(\text{C}\) does not show any peaks between 9.0 − 13.0 ppm, it cannot be a carboxylic acid. Compound \(\text{C}\) is therefore a ketone and Compound \(\text{B}\) is a secondary alcohol.
→ The \(\ce{^{1}H NMR}\) spectrum shows 5 peaks \(\Rightarrow \) 5 hydrogen environments.
→ Chemical shift and splitting patterns information indicate:
1.01 ppm – 1.05 ppm: \(\ce{CH3}\) (next to a \(\ce{CH2}\))
1.65 ppm: \(\ce{CH2}\) (with multiple neighbouring hydrogens)
2.42 ppm: \(\ce{CH2}\) (next to the ketone \(\ce{C=O}\) and a \(\ce{CH2}\))
2.46 ppm: \(\ce{CH2}\) (next to the ketone \(\ce{C=O}\) and a \(\ce{CH3}\))
Compound \(\text{A}\): Alkene
Compound \(\text{B}\): Secondary alcohol
Compound \(\text{C}\): Ketone
Reasoning as follows:
→ Compound \(\text{A}\) is able to undergo an addition reaction to add water across a \(\ce{C=C}\) bond \(\Rightarrow \) Alkene
→ Compound \(\text{B}\) is the product of the above hydration reaction and is therefore an alcohol.
→ The \(\ce{^{13}C\ NMR}\) spectrum of Compound \(\text{A}\) confirms it is an alkene (132 ppm peak corresponding to the \(\ce{C=C}\) atoms). 3 spectrum peaks indicate 3 carbon environments. The molar mass of compound \(\text{A}\) is 84.156 g mol\(^{-1}\) which suggests symmetry within the molecule.
→ The Infrared Spectrum of Compound \(\text{B}\) has a broad peak at approximately 3400 cm\(^{-1}\). This indicates the presence of an hydroxyl group and confirms \(\text{B}\) is an alcohol.
→ Compound \(\text{C}\) is produced by the oxidation of Compound \(\text{B}\) with acidified potassium permanganate.
→ Compound \(\text{C}\) is a carboxylic acid if \(\text{B}\) is a primary alcohol or a ketone if \(\text{B}\) is a secondary alcohol.
→ Since the \(\ce{^{1}H NMR}\) spectrum of \(\text{C}\) does not show any peaks between 9.0 − 13.0 ppm, it cannot be a carboxylic acid. Compound \(\text{C}\) is therefore a ketone and Compound \(\text{B}\) is a secondary alcohol.
→ The \(\ce{^{1}H NMR}\) spectrum shows 5 peaks \(\Rightarrow \) 5 hydrogen environments.
→ Chemical shift and splitting patterns information indicate:
1.01 ppm – 1.05 ppm: \(\ce{CH3}\) (next to a \(\ce{CH2}\))
1.65 ppm: \(\ce{CH2}\) (with multiple neighbouring hydrogens)
2.42 ppm: \(\ce{CH2}\) (next to the ketone \(\ce{C=O}\) and a \(\ce{CH2}\))
2.46 ppm: \(\ce{CH2}\) (next to the ketone \(\ce{C=O}\) and a \(\ce{CH3}\))
The technique illustrated is used to analyse chemical substances in a sample.
What is the technique shown?
\(C\)
By elimination:
→ Although a flame test uses a flame to analyse chemical samples, it does not require a detector, prism, lamp or lens. Hence it is not the analytical method being demonstrated in the above diagram (eliminate A).
→ Mass Spectroscopy is used for organic compounds and requires an electromagnet which is not present in the above diagram (eliminate B).
→ Atomic Absorption Spectroscopy (used for inorganic compounds) is based on the idea that atoms can absorb light at a specific unique wavelength. The above image demonstrates this.
→ Ultraviolet-visible Spectrophotometry: although this uses the same principle as AAS to detect sample concentration, Ultraviolet-visible Spectrophotometry uses a different wavelength of light and requires a different apparatus to the one in the diagram above (eliminate D).
\(\Rightarrow C\)
When performing industrial reductions with \(\mathrm{CO}(\mathrm{g})\), the following equilibrium is of great importance.
\( \ce{2CO(g) \rightleftharpoons CO2(g) + C(s) \quad \quad $K$_{e q} = 10.00 at 1095 K } \)
A 1.00 L sealed vessel at a temperature of 1095 K contains \( \ce{CO(g)} \) at a concentration of 1.10 × 10\(^{-2}\) mol L\(^{-1}\), \(\ce{CO2(g)} \) at a concentration of 1.21 × 10\(^{-3}\) mol L\(^{-1}\), and excess solid carbon.
--- 3 WORK AREA LINES (style=lined) ---
Carbon dioxide gas is added to the system above and the mixture comes to equilibrium. The equilibrium concentrations of \( \ce{CO(g)}\) and \(\ce{CO2(g)} \) are equal. Excess solid carbon is present and the temperature remains at 1095 K.
Calculate the amount (in mol) of carbon dioxide added to the system. (3 marks)
--- 10 WORK AREA LINES (style=lined) ---
| a. | \(Q\) | \(=\dfrac{\ce{[CO2]}}{\ce{[CO]^2}} \) |
| \(=\dfrac{1.21 \times 10^{-3}}{(1.10 \times 10^{-2})^2} \) | ||
| \(=10.0\) |
\(\text{Since}\ \ Q=K_{eq},\ \text{system is in equilibrium.}\)
b. \(0.143\ \text{mol} \)
| a. | \(Q\) | \(=\dfrac{\ce{[CO2]}}{\ce{[CO]^2}} \) |
| \(=\dfrac{1.21 \times 10^{-3}}{(1.10 \times 10^{-2})^2} \) | ||
| \(=10.0\) |
\(\text{Since}\ \ Q=K_{eq},\ \text{system is in equilibrium.}\)
b. \(\ce{\text{Given}\ \ [CO]=[CO2]}, \)
\(K_{eq} =\dfrac{\ce{[CO2]}}{\ce{[CO]^2}} =\dfrac{1}{\ce{[CO]}} = 10.00\)
\(\Rightarrow \ce{[CO] = \dfrac{1}{10.00} = 0.1000 \text{mol L}^{-1}} \)
\(\Rightarrow \ce{[CO2] = 0.1000 \text{mol L}^{-1}} \)
From this point, the change in \(\ce{CO}\) and \(\ce{CO2}\) concentrations can be calculated…
\begin{array} {|l|c|c|c|}
\hline & \ce{2CO(g)} & \ce{CO2(g)} & \ce{C(s)} \\
\hline \text{Initial} & 1.10 \times 10^{-2} & 1.21 \times 10^{-3} & \\
\hline \text{Change} & +0.0890 & +0.0988 & \\
\hline \text{Equilibrium} & \ \ \ 0.1000 & \ \ \ 0.1000 & \\
\hline \end{array}
However, the change in moles of \(\ce{CO2}\) in the system consists of:
\(\ce{n(CO2)\ \text{required to increase}\ [CO] by 0.0988\ \text{mol}\ \ \ \text{(1 litre vessel)}}\)
\(\ce{\text{Formula ratio shows}\ \ CO2:CO = 1\ \text{mol} : 2\ \text{mol}} \)
\(\ce{n(CO2)\ \text{to add to increase}\ [CO2] = 0.0988\ \text{mol}\ \ \ \text{(1 litre vessel)}}\)
\(\ce{n(CO2)_{\text{total to add}} = 0.0988\ \text{mol} + n(CO2\ \text{to make CO)}} \)
\(\ce{n(CO2)\ \text{to add to increase}\ [CO] = \dfrac{0.0890}{2} = 0.0445\ \text{mol}}\)
\(\ce{n(CO2)_{\text{total to add}} = 0.0988 + 0.0445 = 0.143\ \text{mol}} \)
Gases \( \ce{A_2} \) and \( \ce{B_2} \) are placed in a closed container of variable volume, as shown.
The reaction between these substances is as follows.
\( \ce{A2(g) + 2B_2(g) \rightleftharpoons 2AB_2(g) \quad \Delta \textit{H} = -10 \text{kJ mol}^{-1}} \)
The following graph shows changes in the amounts (in mol) of these three substances over time in this container.
--- 4 WORK AREA LINES (style=lined) ---
--- 8 WORK AREA LINES (style=lined) ---
a. Between 6 and 8 minutes:
→ The system is in equilibrium.
→ The horizontal lines of each reactant in the graph indicate that the amount of reactants and products remain constant and hence the forward and reverse reactions are proceeding at the same rate.
b. After 8 minutes \(\ce{AB2}\) is consumed, and \(\ce{A2}\) and \(\ce{B2}\) are produced.
Factor 1:
→ An increase in temperature that decreases the equilibrium constant, \(\text{K}\).
→ In this case, the reaction quotient \(\text{Q}\) will be greater than \(\text{K}\). This will result in \(\ce{AB2}\) being consumed and \(\ce{A2}\) and \(\ce{B2}\) being produced until \(\text{Q}\) approaches \(\text{K}\) and the system reaches equilibrium again.
Factor 2:
→ Increase in volume of the container.
→ This will increase the reaction quotient \(\text{Q}\) while \(\text{K}\) stays the same. Again, this will cause \(\ce{AB2}\) to be consumed and \(\ce{A2}\) and \(\ce{B2}\) to be produced until \(\text{Q}\) approaches \(\text{K}\) and the system reaches equilibrium again.
a. Between 6 and 8 minutes:
→ The system is in equilibrium.
→ The horizontal lines of each reactant in the graph indicate that the amount of reactants and products remain constant and hence the forward and reverse reactions are proceeding at the same rate.
b. After 8 minutes \(\ce{AB2}\) is consumed, and \(\ce{A2}\) and \(\ce{B2}\) are produced.
Factor 1:
→ An increase in temperature that decreases the equilibrium constant, \(\text{K}\).
→ In this case, the reaction quotient \(\text{Q}\) will be greater than \(\text{K}\). This will result in \(\ce{AB2}\) being consumed and \(\ce{A2}\) and \(\ce{B2}\) being produced until \(\text{Q}\) approaches \(\text{K}\) and the system reaches equilibrium again.
Factor 2:
→ Increase in volume of the container.
→ This will increase the reaction quotient \(\text{Q}\) while \(\text{K}\) stays the same. Again, this will cause \(\ce{AB2}\) to be consumed and \(\ce{A2}\) and \(\ce{B2}\) to be produced until \(\text{Q}\) approaches \(\text{K}\) and the system reaches equilibrium again.
Copper(\(\text{II}\)) ions \( \ce{(Cu^{2+})} \) form a complex with lactic acid \( \ce{(C3H6O3)} \), as shown in the equation.
\( \ce{Cu^{2+}(aq)} + \ce{2C3H6O3(aq)} \rightleftharpoons \Bigl[\ce{Cu(C3H6O3)2\Bigr]^{2+}(aq)} \)
This complex can be detected by measuring its absorbance at 730 nm. A series of solutions containing known concentrations of \( \Bigl[\ce{Cu(C3H6O3)_2\Big]^{2+}} \) were prepared, and their absorbances measured.
| \( Concentration \ of \Bigl[\ce{Cu(C3H6O3)_2\Bigr]^{2+}} \) \( \text{(mol L}^{-1}) \) | \( Absorbance \) |
| 0.000 | 0.00 |
| 0.010 | 0.13 |
| 0.020 | 0.28 |
| 0.030 | 0.43 |
| 0.040 | 0.57 |
| 0.050 | 0.72 |
| \( Species \) | \( Initial \ Concentration\) \( (\text{mol L}^{-1}) \) |
| \( \ce{Cu^{2+}} \) | 0.056 |
| \( \ce{C3H6O3} \) | 0.111 |
When the solution reached equilibrium, its absorbance at 730 nm was 0.66.
You may assume that under the conditions of this experiment, the only species present in the solution are those present in the equation above, and that \( \Bigl[ \ce{Cu(C3H6O3)_2\Bigr]^{2+}} \) is the only species that absorbs at 730 nm.
With the support of a line graph, calculate the equilibrium constant for the reaction. (7 marks)
--- 11 WORK AREA LINES (style=lined) ---
\(\text{From graph:}\)
\(\text{0.66 absorbance}\ \Rightarrow\ \ \Big[\bigl[\ce{Cu(C3H6O3)2\bigr]^{2+}\Big]} = 0.046\ \text{mol L}^{-1} \)
\begin{array} {|l|c|c|c|}
\hline & \ce{Cu^{2+}} & \ce{2C3H6O3(aq)} & \ce{\big[Cu(C3H6O3)2\big]^{2+}(aq)} \\
\hline \text{Initial} & \ \ \ \ 0.056 & \ \ \ \ 0.111 & 0 \\
\hline \text{Change} & -0.046 & -0.092 & \ \ \ +0.046 \\
\hline \text{Equilibrium} & \ \ \ \ 0.010 & \ \ \ \ 0.019 & \ \ \ \ \ \ 0.046 \\
\hline \end{array}
| \(K_{eq}\) | \(=\dfrac{\ce{\Big[\big[Cu(C3H6O3)2\big]^{2+}\Big]}}{\ce{\big[Cu^{2+}\big]\big[C3H6O3\big]^2}}\) | |
| \(=\dfrac{0.046}{0.010 \times 0.019^2}\) | ||
| \(=1.3 \times 10^4\) |
The following graph shows the solubility of some alkan-1-ols in water at 20°C.
Explain the relationship between the trend shown in the graph and the relevant intermolecular forces. (3 marks)
--- 8 WORK AREA LINES (style=lined) ---
→ The graph shows a non-linear relationship with the following clear trend, as the molar mass increases, solubility decreases.
→ When molar mass increases, the chain length of a molecule increases. Hence, as the chain length of alkan-1-ols increase, their solubility in water decreases.
→ Shorter chain alcohols dissolve more readily in water. This is due to the formation of hydrogen bonds between the hydroxyl group of the alcohol and water molecules.
→ However, as the chain length of alkan-1-ols increase, the dispersion forces between the alkyl groups become stronger, decreasing their solubility.
→ The graph shows a non-linear relationship with the following clear trend, as the molar mass increases, solubility decreases.
→ When molar mass increases, the chain length of a molecule increases. Hence, as the chain length of alkan-1-ols increase, their solubility in water decreases.
→ Shorter chain alcohols dissolve more readily in water. This is due to the formation of hydrogen bonds between the hydroxyl group of the alcohol and water molecules.
→ However, as the chain length of alkan-1-ols increase, the dispersion forces between the alkyl groups become stronger, decreasing their solubility.
my solution
→ The graph shows a non-linear relationship with the following clear trend, as the molar mass increases, solubility decreases.
→ When molar mass increases, the chain length of a molecule increases. In alkan-1-ols this increases the length of their carbon backbone, increasing their non-polar nature (increased dispersion forces), thus solubility in polar solvents (eg: water) decreases
→ Shorter chain alcohols dissolve more readily in water. This is due to the formation of hydrogen bonds between the hydroxyl group of the alcohol and water molecules and the comparatively polar nature of the molecule compared top long-chained alkan-1-ols
→ However, as the chain length of alkan-1-ols increase, the dispersion forces between the alkyl groups become stronger and mitigate the polarity of the hydroxyl group, decreasing their solubility.
Alkene \(\ce{Q}\) undergoes an addition reaction with chlorine gas to form compound \(\ce{R}\).
--- 4 WORK AREA LINES (style=lined) ---
--- 8 WORK AREA LINES (style=lined) ---
a. Chemical test for an alkene
→ Prepare a sample of alkene \(\ce{Q}\) in a clean test tube.
→ Add a few drops of bromine water to the sample.
→ The bromine water will be decolourised if \(\ce{Q}\) is an alkene.
Other correct answers could include:
→ The addition of potassium permanganate will also be decolourised by \(\ce{Q}\) if it is an alkene.
b. Molecular ion is present at m/z = 114
Atomic mass of \(\text{C}\) (in compound \(\ce{R}\)) = 32% × 114 = 36
\(\text{C}\) atoms in 1 molecule of R = 36 ÷ 12 = 3
Mass (non-\(\text{C}\)) = 114 – 36 = 78
\(\Rightarrow\) Two atoms of \(\ce{Cl}\) are in compound \(\ce{R}\)
\(\therefore\) \(\ce{R}\) has the formula \(\ce{C3H6Cl2}\), and structure:
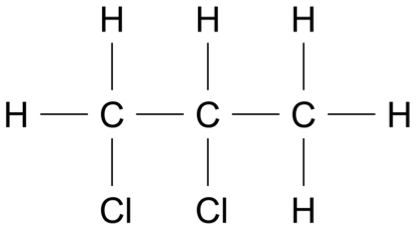
a. Chemical test for an alkene
→ Prepare a sample of alkene \(\ce{Q}\) in a clean test tube.
→ Add a few drops of bromine water to the sample.
→ The bromine water will be decolourised if \(\ce{Q}\) is an alkene.
Other correct answers could include:
→ The addition of potassium permanganate will also be decolourised by \(\ce{Q}\) if it is an alkene.
b. Molecular ion is present at m/z = 114
Atomic mass of \(\text{C}\) (in compound \(\ce{R}\)) = 32% × 114 = 36
\(\text{C}\) atoms in 1 molecule of R = 36 ÷ 12 = 3
Mass (non-\(\text{C}\)) = 114 – 36 = 78
\(\Rightarrow\) Two atoms of \(\ce{Cl}\) are in compound \(\ce{R}\)
\(\therefore\) \(\ce{R}\) has the formula \(\ce{C3H6Cl2}\), and structure:

A student has been asked to produce 185 mL of ethanol (MM = 46.068 g mol\(^{-1} \)) by fermenting glucose using yeast, as shown in the equation.
\( \ce{C6H12O6(aq)} \rightarrow \ce{2C2H5OH(aq)} + \ce{2CO2(g)} \)
Given that the density of ethanol is 0.789 g mL\(^{-1} \), calculate the volume of carbon dioxide gas produced at 310 K and 100 kPa. (4 marks)
--- 7 WORK AREA LINES (style=lined) ---
\(81.7\ \text{L}\)
\(\text{Density}(\rho)\ = \dfrac{\text{m}}{\text{V}}\ \ \Rightarrow\ \ \text{m} = \rho \times\ \text{V} \)
\(\text{m(ethanol)}\ = 0.789 \times 185 = 146\ \text{g} \)
\(\text{n(ethanol)}\ = \dfrac{\text{m}}{\text{MM}} = \dfrac{146}{46.068} = 3.17\ \text{mol} \)
| \(\text{V}\) | \(=\dfrac{\text{n} RT}{P} \) | |
| \(=\dfrac{3.17 \times 8.314 \times 310}{100}\) | ||
| \(=81.7\ \text{L}\) |
A student used the apparatus shown to investigate the combustion of octan-1-ol.
The following results were obtained by the student.
| Mass of water heated | = 205 g |
| Initial temperature of water | = 23.7°C |
| Final temperature of water | = 60.4°C |
The following data are given.
| Molar enthalpy of combustion of octan-1-ol | = – 5294 kJ mol\(^{-1} \) |
| Molar mass of octan-1-ol | = 130.23 g kJ mol\(^{-1}\) |
--- 8 WORK AREA LINES (style=lined) ---
--- 4 WORK AREA LINES (style=lined) ---
a. 0.774 grams
b. Advantage of biofuel vs fossil fuel
→ Combustion of biofuels derived from plants will have a lower greenhouse impact as the carbon dioxide released during combustion will replace that used in photosynthesis, unlike fossil fuels.
Other answers could include:
→ Biofuels are biodegradable and therefore pose a much reduced environmental threat than fossil fuels which are not.
→ Biofuels are more sustainable than fossil fuels as they are produced from renewable resources.
a. Find the heat absorbed by the water ((\q\)):
| \(q\) | \(=mc \Delta T \) | |
| \(=205 \times 4.18 \times 36.7 \) | ||
| \(=31\ 488.23\ \text{J}\) | ||
| \(=31.448\ \text{kJ}\) |
\(\text{octan-1-ol}\ \Rightarrow \ce{C8H18O} \)
\(\ce{MM(C8H18O) = 12.01 \times 8 + 1.008 \times 18 + 16 = 130.224} \)
| \(\ce{n\text{(octan-1-ol)}}\) | \(= \dfrac{-31.448\ \text{kJ}}{-5294\ \text{kJ mol}^{-1}} \) | |
| \(=5.94 \times 10^{-3}\ \text{mol} \) |
| \(\ce{m\text{(octan-1-ol)}}\) | \(= 5.94 \times 10^{-3} \times 130.224 \) | |
| \(= 0.774\ \text{g} \) |
b. Advantage of biofuel vs fossil fuel
→ Combustion of biofuels derived from plants will have a lower greenhouse impact as the carbon dioxide released during combustion will replace that used in photosynthesis, unlike fossil fuels.
Other answers could include:
→ Biofuels are biodegradable and therefore pose a much reduced environmental threat than fossil fuels which are not.
→ Biofuels are more sustainable than fossil fuels as they are produced from renewable resources.
The hydrogen oxalate ion \( \ce{(HC2O4^{-})} \) is classified as amphiprotic.
Describe, using chemical equations, how this ion is amphiprotic. (2 marks)
--- 4 WORK AREA LINES (style=lined) ---
→ \(\ce{HC2O4−}\) is amphiprotic because it can either accept or donate an \(\ce{H+}\) as shown in the following equations:
\(\ce{HC2O4−(aq) + H+(aq) \rightleftharpoons H2C2O4(aq)}\)
\(\ce{HC2O4−(aq) + OH−(aq) \rightleftharpoons H2O(l) + C2O4^{2−}(aq)}\)
→ \(\ce{HC2O4−}\) is amphiprotic because it can either accept or donate an \(\ce{H+}\) as shown in the following equations:
\(\ce{HC2O4−(aq) + H+(aq) \rightleftharpoons H2C2O4(aq)}\)
\(\ce{HC2O4−(aq) + OH−(aq) \rightleftharpoons H2O(l) + C2O4^{2−}(aq)}\)
The pH of two solutions, \(\text{X}\) and \(\text{Y}\), were measured before and after 10 drops of concentrated \( \ce{NaOH} \) was added to each.
Explain the pH changes that occurred in solutions \(\text{X}\) and \(\text{Y}\). (3 marks)
--- 8 WORK AREA LINES (style=lined) ---
→ The diagram shows that the pH of solution \(\text{X}\) changes significantly with the introduction of the base \(\ce{NaOH}\), whereas the pH of solution \(\text{Y}\) only shows a small change in pH. This indicates that solution \(\text{Y}\) contains a buffer while solution \(\text{X}\) does not.
→ When \(\ce{NaOH}\) was added to solution \(\text{X}\), the addition of \(\ce{OH-}\) ions caused the increase in pH \(\ce{(pH = −log10 [H3O+]).} \)
→ In contrast, the \(\ce{OH-}\) ions react with the buffer solution in solution \(\text{Y}\). This has the effect of minimising the change in \(\ce{[H3O+]}\) and therefore pH.
→ The diagram shows that the pH of solution \(\text{X}\) changes significantly with the introduction of the base \(\ce{NaOH}\), whereas the pH of solution \(\text{Y}\) only shows a small change in pH. This indicates that solution \(\text{Y}\) contains a buffer while solution \(\text{X}\) does not.
→ When \(\ce{NaOH}\) was added to solution \(\text{X}\), the addition of \(\ce{OH-}\) ions caused the increase in pH \(\ce{(pH = −log10 [H3O+]).} \)
→ In contrast, the \(\ce{OH-}\) ions react with the buffer solution in solution \(\text{Y}\). This has the effect of minimising the change in \(\ce{[H3O+]}\) and therefore pH.
Explain how the following substances would be classified under the Arrhenius and Brønsted-Lowry definitions of acids. Support your answer with relevant equations. (4 marks)
--- 12 WORK AREA LINES (style=lined) ---
→ Acids are defined by Arrhenius as hydrogen-containing compounds that dissociate in water to give \(\ce{H+}\) ions.
→ \(\ce{HCl(aq)}\) produces \(\ce{H+}\) ions in water and therefore qualifies within Arrhenius’ definition of an acid.
\(\ce{HCl(aq) \rightarrow H+(aq) + Cl−(aq)}\)
→ The salt \(\ce{NH4Cl}\) would not be recognised as an acid with Arrhenius’ definition, since the predominant ions present in aqueous solution are ammonium and chloride.
→ The Brønsted−Lowry theory states that acids are proton donors. \(\ce{HCl(aq)}\) is a proton donor and therefore also qualifies as a Brønsted−Lowry acid.
→ In contradiction to Arrhenius, ammonium chloride \(\ce{(NH4Cl)}\) is also classified as a Brønsted−Lowry acid. This is due to the ammonium ion donating a proton to water to form a hydronium ion.
\(\ce{NH4+(aq) + H2O(l) \rightleftharpoons NH3(aq) + H3O+(aq)}\)
→ Acids are defined by Arrhenius as hydrogen-containing compounds that dissociate in water to give \(\ce{H+}\) ions.
→ \(\ce{HCl(aq)}\) produces \(\ce{H+}\) ions in water and therefore qualifies within Arrhenius’ definition of an acid.
\(\ce{HCl(aq) \rightarrow H+(aq) + Cl−(aq)}\)
→ The salt \(\ce{NH4Cl}\) would not be recognised as an acid with Arrhenius’ definition, since the predominant ions present in aqueous solution are ammonium and chloride.
→ The Brønsted−Lowry theory states that acids are proton donors. \(\ce{HCl(aq)}\) is a proton donor and therefore also qualifies as a Brønsted−Lowry acid.
→ In contradiction to Arrhenius, ammonium chloride \(\ce{(NH4Cl)}\) is also classified as a Brønsted−Lowry acid. This is due to the ammonium ion donating a proton to water to form a hydronium ion.
\(\ce{NH4+(aq) + H2O(l) \rightleftharpoons NH3(aq) + H3O+(aq)}\)
The pH of a solution changes from 8 to 5.
What happens to the concentration of hydrogen ions during this change of pH?
\(C\)
→ Each increase/decrease of pH by a magnitude of 1 represents a change in \(\ce{[H+]}\) of a factor of 10.
→ Therefore, concentration change when pH moves from 8 to 5 = 10 × 3 = 1000 (increase).
\(\Rightarrow C\)
The diagram shows apparatus that is used to investigate the interaction between the magnetic field produced by a coil and two copper rings \(X\) and \(Y\), when each is placed at position \(P\), as shown. Ring \(X\) is a complete circular ring, and a small gap has been cut in ring \(Y\). Each of the rings has a cross-sectional area of 4 × 10\(^{-4}\) m². The power supply connected to the coil produces an increasing current through the coil in the direction shown, when the switch is turned on. This produces a magnetic field at \(P\) that varies as shown in the graph. --- 7 WORK AREA LINES (style=lined) --- --- 4 WORK AREA LINES (style=lined) --- --- 4 WORK AREA LINES (style=lined) --- a. Between 0 – 0.03 seconds: → The magnetic field strength at point \(P\) is increasing at a constant rate. → Thus, ring \(X\) experiences a change in flux, which causes an EMF to be induced across the ring (Faraday’s Law) → This induced EMF causes a current to flow through ring \(X\) that will interact in the external magnetic field to reduce the change in flux that created it (Lenz’s Law). → Thus, a clockwise current will run through the ring, creating a north pole towards the solenoid, causing the ring to have a repulsive force away from the solenoid acting on it. Between 0.03 – 0.05 seconds: → There is no change in flux through the ring due to there being a constant magnetic field. → Therefore, there is no induced EMF or induced current. → Therefore, there is no force acting on the ring. b.i. Ring \(Y\) behaviour when placed at \(P\): → Ring \(Y\) will experience the same change in flux and hence the same induced EMF as ring \(X\) within 0 – 0.03 seconds. → However, due to the gap in ring \(Y\) (i.e. there is no closed circuit), no induced current will be able to flow through the ring. → Hence, there is no force exerted on ring \(Y\). ii. \(\varepsilon = 8 \times 10^{-5}\ \text{V}\) a. Between 0 – 0.03 seconds: → The magnetic field strength at point \(P\) is increasing at a constant rate. → Thus, ring \(X\) experiences a change in flux, which causes an EMF to be induced across the ring (Faraday’s Law) → This induced EMF causes a current to flow through ring \(X\) that will interact in the external magnetic field to reduce the change in flux that created it (Lenz’s Law). → Thus, a clockwise current will run through the ring, creating a north pole towards the solenoid, causing the ring to have a repulsive force away from the solenoid acting on it. Between 0.03 – 0.05 seconds: → There is no change in flux through the ring due to there being a constant magnetic field. → Therefore, there is no induced EMF or induced current. → Therefore, there is no force acting on the ring. b.i. Ring \(Y\) behaviour when placed at \(P\): → Ring \(Y\) will experience the same change in flux and hence the same induced EMF as ring \(X\) within 0 – 0.03 seconds. → However, due to the gap in ring \(Y\) (i.e. there is no closed circuit), no induced current will be able to flow through the ring. → Hence, there is no force exerted on ring \(Y\).
b.ii.
\(\varepsilon\)
\(=N\dfrac{\Delta \phi}{\Delta t}\)
\(=N \dfrac{A \Delta B}{\Delta t}\)
\(= 1 \times \dfrac{4 \times 10^{-4} \times (6 \times 10^{-3}-0)}{0.03-0}\)
\(=8 \times 10^{-5}\ \text{V}\)
The graph shows the increase in the global yield of wheat from 1800-2020. Genetically modified organisms (GMOs) are not currently used to grow wheat commercially.
What row in the table correctly identifies biotechnologies that have contributed to the increase in wheat yields and could be adapted to enhance commercial production in the future?
| Past (until 1960) | Present (1960-2023) | Future (2023 onward) | |
| A. | Selective breeding Embryo transfer |
GMO production Gene sequencing |
CRISPR Recombinant DNA technologies |
| B. | Selective breeding Embryo transfer |
Selective breeding Gene sequencing |
CRISPR Stem cell engineering |
| C. | Selective breeding Hybridisation | Artificial insemination Recombinant DNA technologies | CRISPR Stem cell engineering |
| D. | Selective breeding Hybridisation | Selective breeding Gene sequencing |
CRISPR Recombinant DNA technologies |
\(D\)
→ Wheat does not have an embryo and only animals can be artificially inseminated.
\(\Rightarrow D\)
Cattle have been domesticated by humans for approximately 10 000 years. Many biotechnologies have been employed in the farming of cattle. The table shows examples of the application of these biotechnologies. → Biotechnologies can increase, decrease or maintain the size of the gene pool in populations and species, particularly in the case of cattle which have been subject to a range of biotechnologies. → Selective breeding, which decreases biodiversity, has been used for hundreds of years by farmers who oversee the reproduction of cattle with favourable characteristics, such as females who produce the most milk. → Artificial insemination, which typically reduces biodiversity, allows a single bull to sire many offspring. This process breeds out certain characteristics of cattle, reducing the diversity of the species. However, in certain circumstances, the gene pool of specific communities can be diversified through the introduction of new alleles. → Whole cattle cloning reduces biodiversity by making cloned organisms that are identical genotypes to the parent. As the success rate is low and cloned animals are infertile, this does not have the potential to have a large impact on biodiversity. → Hybridisation generally increases biodiversity by naturally mating two different cattle species and in the process, introducing genes not originally present. → Hybridisation can however also reduce biodiversity if cattle hybrids are then selectively bred in preference to the original breeds. → Transgenic organisms are produced where new alleles are artificially introduced into the species, increasing biodiversity. As this process is expensive and not widespread, it will not have a large effect on biodiversity. → In summary, the most wide spread and influential biotechnologies have the overall effect of decreasing the biodiversity of cattle. → Biotechnologies can increase, decrease or maintain the size of the gene pool in populations and species, particularly in the case of cattle which have been subject to a range of biotechnologies. → Selective breeding, which decreases biodiversity, has been used for hundreds of years by farmers who oversee the reproduction of cattle with favourable characteristics, such as females who produce the most milk. → Artificial insemination, which typically reduces biodiversity, allows a single bull to sire many offspring. This process breeds out certain characteristics of cattle, reducing the diversity of the species. However, in certain circumstances, the gene pool of specific communities can be diversified through the introduction of new alleles. → Whole cattle cloning reduces biodiversity by making cloned organisms that are identical genotypes to the parent. As the success rate is low and cloned animals are infertile, this does not have the potential to have a large impact on biodiversity. → Hybridisation generally increases biodiversity by naturally mating two different cattle species and in the process, introducing genes not originally present. → Hybridisation can however also reduce biodiversity if cattle hybrids are then selectively bred in preference to the original breeds. → Transgenic organisms are produced where new alleles are artificially introduced into the species, increasing biodiversity. As this process is expensive and not widespread, it will not have a large effect on biodiversity. → In summary, the most wide spread and influential biotechnologies have the overall effect of decreasing the biodiversity of cattle.
Biotechnology
Example
Selective breeding
The offspring of highest milk producing female cows were retained and over time cows that produced more milk were bred, leading to dairy breeds.
Artificial insemination
An American bull holds the current record for artificial insemination. He produced 2.4 million units of semen and has sired cattle in 50 countries.
Whole organism cloning
The success rate of cloning cattle is low. There are currently 30-40 cloned cattle in Australia. They are not used commercially.
Hybridisation
There are two species of domestic cattle, Bos taurus and Bos indicus. They can be hybridised to breed cattle with characteristics of both species.
Transgenic organisms
The first transgenic cow produced human serum albumin in its milk. The use of transgenic cattle is not widespread.
With reference to the table, evaluate the effect of biotechnologies on the biodiversity of cattle. (5 marks)
The mountain pygmy possum (Burramys parvus) is restricted to four regions in Australia's alpine zone. The species is listed as critically endangered with less than 2000 adults remaining. The range of the mountain pygmy possum has contracted due to a gradually warming climate. Loss and degradation of these habitats have affected local populations. The graph shows changes in the Mt Buller population following recent bushfires and the introduction of male pygmy possums from Mt Bogong. Evaluate how bushfires and the introduction of males from other locations have affected the population size and gene pool of the Mt Buller pygmy possum population. (7 marks) --- 18 WORK AREA LINES (style=lined) --- → Environmental factors can play extremely large roles in the population and hence gene pool of the pygmy possums in Mt Buller. → This can be seen especially between 1996-2007, where three bushfires throughout those years caused the population to drop from 90 to less than 10. This is because bushfires not only kill individual possums, but also destroy their habitat, reducing shelter, food and water sources for the survivors. → This catastrophic drop in population significantly reduced the alleles present in the Mt Buller gene pool. The “new” survivor gene pool would see some alleles potentially disappear while others appear more frequently in individuals. → This may have led to genetic drift (bottleneck effect) in this population where the low diversity of alleles in the population led to a further reduction in the population between 2002–2007. → To counteract this reduction in population and gene pool size, 6 male possums were introduced from a nearby population in 2007. This was done again in 2012, and despite there being another bushfire in 2011, the population increased to 150 by 2015. → The introduction of these males in 2007 almost doubled the known population at the time, but just as importantly diversified the gene pool, an effect helped by the isolation of the two populations. → This increase in genetic diversity also improves a species’ ability to adapt to the drier and hotter climates being experienced. → It is evident that both the bushfires and the introduction of males played different but very important roles in the population size and gene pool of the Mt Buller pygmy possum population. → Environmental factors can play extremely large roles in the population and hence gene pool of the pygmy possums in Mt Buller. → This can be seen especially between 1996-2007, where three bushfires throughout those years caused the population to drop from 90 to less than 10. This is because bushfires not only kill individual possums, but also destroy their habitat, reducing shelter, food and water sources for the survivors. → This catastrophic drop in population significantly reduced the alleles present in the Mt Buller gene pool. The “new” survivor gene pool would see some alleles potentially disappear while others appear more frequently in individuals. → This may have led to genetic drift (bottleneck effect) in this population where the low diversity of alleles in the population led to a further reduction in the population between 2002–2007. → To counteract this reduction in population and gene pool size, 6 male possums were introduced from a nearby population in 2007. This was done again in 2012, and despite there being another bushfire in 2011, the population increased to 150 by 2015. → The introduction of these males in 2007 almost doubled the known population at the time, but just as importantly diversified the gene pool, an effect helped by the isolation of the two populations. → This increase in genetic diversity also improves a species’ ability to adapt to the drier and hotter climates being experienced. → It is evident that both the bushfires and the introduction of males played different but very important roles in the population size and gene pool of the Mt Buller pygmy possum population.
Describe a named genetic technology and its use in a medical application. (4 marks) → A polymerase chain reaction (PCR) is a process which allows scientists to replicate billions of copies of a specific gene. → The PCR process involves denaturing a DNA sample by heating it to around 98°C. The sample is then cooled to 60°C where DNA primers, polymerase enzymes and free nucleotides are added, making a copy of the original sample. → By repeating this process, the amount of copies acquired will increase exponentially. → This process is used in COVID-19 testing centres. → An individual’s DNA is swabbed and a PCR test is used to multiply it for COVID testing. PCR can also be used in other medical scenarios where gene cloning is required, such as in the production of mRNA vaccines. Other answers could include → IVF. → Recombinant DNA in bacteria to produce insulin. → A polymerase chain reaction (PCR) is a process which allows scientists to replicate billions of copies of a specific gene. → The PCR process involves denaturing a DNA sample by heating it to around 98°C. The sample is then cooled to 60°C where DNA primers, polymerase enzymes and free nucleotides are added, making a copy of the original sample. → By repeating this process, the amount of copies acquired will increase exponentially. → This process is used in COVID-19 testing centres. → An individual’s DNA is swabbed and a PCR test is used to multiply it for COVID testing. PCR can also be used in other medical scenarios where gene cloning is required, such as in the production of mRNA vaccines. Other answers could include → IVF. → Recombinant DNA in bacteria to produce insulin.
Tetanus vaccines were introduced in 1953 resulting in reduced case numbers. The majority of recorded cases occurred in people aged 65 and over. The graph shows the vaccination schedule for tetanus. Assess the use of vaccinations and the vaccination schedule. Use the data provided to support your answer. (5 marks) → Vaccines are an excellent tool to reduce the severity and spread of certain infectious diseases. → This is because vaccines contain dead or inactive versions of a pathogen, which then stimulates the body to fight it and store memory B cells with the associated antibodies. → This can be clearly seen on the graph after the first and second injections of the vaccine, where the first injection is the initial exposure while the second injection has a much larger production of antibodies due to the memory B cells being activated. This is the basis of immunity. → This effect is compounded each time with the the length of immunity increasing after each dose. → The vaccination schedule is designed so that a new dose is scheduled at the time a person following the recommended schedule would become non-immune. → The general trend in the graph/data suggest that the increase in cases of people over 65 is likely due to a long period elapsing since their last booster. → In summary, if an individual on the recommended vaccination schedule is exposed to the tetanus virus after 6 months of age, they would have already built up an adequate immunity to be able to fight the tetanus bacteria. → This effect makes vaccines especially effective in stopping the spread and severity of infectious diseases such as tetanus. → Vaccines are an excellent tool to reduce the severity and spread of certain infectious diseases. → This is because vaccines contain dead or inactive versions of a pathogen, which then stimulates the body to fight it and store memory B cells with the associated antibodies. → This can be clearly seen on the graph after the first and second injections of the vaccine, where the first injection is the initial exposure while the second injection has a much larger production of antibodies due to the memory B cells being activated. This is the basis of immunity. → This effect is compounded each time with the the length of immunity increasing after each dose. → The vaccination schedule is designed so that a new dose is scheduled at the time a person following the recommended schedule would become non-immune. → The general trend in the graph/data suggest that the increase in cases of people over 65 is likely due to a long period elapsing since their last booster. → In summary, if an individual on the recommended vaccination schedule is exposed to the tetanus virus after 6 months of age, they would have already built up an adequate immunity to be able to fight the tetanus bacteria. → This effect makes vaccines especially effective in stopping the spread and severity of infectious diseases such as tetanus.
Organisms use various mechanisms to maintain their internal environment within tolerance limits. Explain TWO adaptations in plants that help to maintain water balance. (4 marks) --- 8 WORK AREA LINES (style=lined) --- Answers could include two of the following: → Leaves are covered in a waxy cuticle on their surface which reduces water loss. Plants that live in hotter and/or dryer areas will have thicker waxy cuticles. → Stomata are holes on the underside of the leaf which allow water to leave the plant via the process of transpiration. Stomata are able to close when the plant is exposed to higher temperatures which reduces transpiration. → Many plants also have sunken stomata which reduces transpiration. → A plant’s roots are able to absorb water from the ground, however the depths of these roots is dependant on the plant’s environment. Some plants such as cacti will have a shallow root system to absorb overnight condensation, while other plants have deeper root systems which allow them to reach the lower water table. Answers could include two of the following: → Leaves are covered in a waxy cuticle on their surface which reduces water loss. Plants that live in hotter and/or dryer areas will have thicker waxy cuticles. → Stomata are holes on the underside of the leaf which allow water to leave the plant via the process of transpiration. Stomata are able to close when the plant is exposed to higher temperatures which reduces transpiration. → Many plants also have sunken stomata which reduces transpiration. → A plant’s roots are able to absorb water from the ground, however the depths of these roots is dependant on the plant’s environment. Some plants such as cacti will have a shallow root system to absorb overnight condensation, while other plants have deeper root systems which allow them to reach the lower water table.
Organisms use various mechanisms to maintain their internal environment within tolerance limits. Outline a physiological adaptation in endotherms which assists in maintaining their internal environment. (2 marks) → Endotherms have developed the ability to change the diameter of their blood vessels to be able to maintain a stable internal temperature. → If their internal temperature increases the blood vessels will dilate (vasodilation) which will cause warm blood to flush through and heat will radiate from the skin. → If their internal temperature drops the blood vessels will constrict (vasoconstriction) which will reduce the heat radiating from the skin and reduce heat loss. Other answers could include → Sweating to maintain temperature. → Shivering/goosebumps to maintain temperature. → Endotherms have developed the ability to change the diameter of their blood vessels to be able to maintain a stable internal temperature. → If their internal temperature increases the blood vessels will dilate (vasodilation) which will cause warm blood to flush through and heat will radiate from the skin. → If their internal temperature drops the blood vessels will constrict (vasoconstriction) which will reduce the heat radiating from the skin and reduce heat loss. Other answers could include → Sweating to maintain temperature. → Shivering/goosebumps to maintain temperature.
--- 5 WORK AREA LINES (style=lined) --- --- 5 WORK AREA LINES (style=lined) --- a. Viral vs Bacterial Pathogen → Bacteria are single celled organisms and can reproduce on their own, outside of a host. → A virus is simply a protein coating around genetic material that can only reproduce via a host, using ribosomes to make copies of itself. Other answers could include: → Bacteria contain plasmids while viruses do not. → Bacteria contain a cell membrane and flagella while viruses are just a protein coat. → The length of a bacterium is 1–10 \(\mu\)m, much larger than the length of a virus which is typically between 0.05–0.1 \(\mu\)m. b. Experimental procedure: → Collect diarrhoea samples from infected individuals and make stool cultures. → If the disease is bacterial, the bacteria will be able to grow and multiply but if it is viral no growth will occur. Other answers could include → Using PCR to multiply DNA within the sample then using antigen testing to determine if the DNA is viral or bacterial in nature. a. Viral vs Bacterial Pathogen → Bacteria are single celled organisms and can reproduce on their own, outside of a host. → A virus is simply a protein coating around genetic material that can only reproduce via a host, using ribosomes to make copies of itself. Other answers could include: → Bacteria contain plasmids while viruses do not. → Bacteria contain a cell membrane and flagella while viruses are just a protein coat. → The length of a bacterium is 1–10 \(\mu\)m, much larger than the length of a virus which is typically between 0.05–0.1 \(\mu\)m. b. Experimental procedure: → Collect diarrhoea samples from infected individuals and make stool cultures. → If the disease is bacterial, the bacteria will be able to grow and multiply but if it is viral no growth will occur. Other answers could include → Using PCR to multiply DNA within the sample then using antigen testing to determine if the DNA is viral or bacterial in nature.
Air pollution has been linked to a variety of non-infectious neurological (brain) disorders. Some of the symptoms include memory loss, cognitive decline and impaired movement and coordination. 500 people from each of three major cities were surveyed and were monitored and tested for a period of 12 months. Each group included males and females aged between 20 and 50 years of age. The results after 12 months were as follows: Evaluate the method used in this epidemiological study in determining a link between air pollution and the symptoms. (7 marks) --- 16 WORK AREA LINES (style=lined) --- → This study is not an effective study for validating a link between air pollution and cognitive disorders due to a variety of issues with the study’s reliability, validity and accuracy. → While the study does have a reasonable sample size and uses 3 different cities, numerous important factors are not specified. These include risk factors such as age, sex, ethnicity and occupation of the participants in each city. → The 12 month timeframe of the study may not be long enough for symptoms to develop. → The study also does not signify the type and degree of symptoms that were experienced. → A non-city comparison where air pollution is low would provide good baseline data against which the data from city participants could be compared. → The study does also not take into account other important demographic factors, such as socioeconomic status and geography within a city. The proximity to industry of an individual’s workplace or residence would be a particularly important risk factor to consider. → Cities should be chosen that have different levels of air pollution in order to look for general trends in the data, such as more exposure to air pollution corresponds to a greater number of symptoms. There is no evidence that this is part of the study design. → This is not a valid nor reliable test in determining a link between air pollution and neurological disorders. Adding prior medical/family history and data relating to the testing centres/cities as well as controlling more variables will lead to a fairer test. → This study is not an effective study for validating a link between air pollution and cognitive disorders due to a variety of issues with the study’s reliability, validity and accuracy. → While the study does have a reasonable sample size and uses 3 different cities, numerous important factors are not specified. These include risk factors such as age, sex, ethnicity and occupation of the participants in each city. → The 12 month timeframe of the study may not be long enough for symptoms to develop. → The study also does not signify the type and degree of symptoms that were experienced. → A non-city comparison where air pollution is low would provide good baseline data against which the data from city participants could be compared. → The study does also not take into account other important demographic factors, such as socioeconomic status and geography within a city. The proximity to industry of an individual’s workplace or residence would be a particularly important risk factor to consider. → Cities should be chosen that have different levels of air pollution in order to look for general trends in the data, such as more exposure to air pollution corresponds to a greater number of symptoms. There is no evidence that this is part of the study design. → This is not a valid nor reliable test in determining a link between air pollution and neurological disorders. Adding prior medical/family history and data relating to the testing centres/cities as well as controlling more variables will lead to a fairer test.
Malaria is a potentially fatal infectious disease that is spread to humans by infected mosquitoes. Scientists investigated the behaviour of 20 mosquitoes for an hour in each of the four containers shown. Aim: To determine if wearing clean clothing reduces the transmission of malaria. Assume infected mosquitoes that land on clothing transmit malaria. --- 3 WORK AREA LINES (style=lined) --- --- 6 WORK AREA LINES (style=lined) --- a. Dependant Variable: Number of mosquitos that land on the clothing. Controlled Variable: Number of mosquitos in each container. Alternative controlled variable: size of container b. Data results show: → Wearing clean clothing, on average, reduces the number of mosquitos that land on clothes. → This effect occurs for both infected and uninfected mosquitos. → Conclusion: wearing clean clothing will reduce the transmission of malaria, as infected mosquitos will land on clean clothing less often than on clothing already worn for a day. a. Dependant Variable: Number of mosquitos that land on the clothing. Controlled Variable: Number of mosquitos in each container. Alternative controlled variable: size of container b. Data results show: → Wearing clean clothing, on average, reduces the number of mosquitos that land on clothes. → This effect occurs for both infected and uninfected mosquitos. → Conclusion: wearing clean clothing will reduce the transmission of malaria, as infected mosquitos will land on clean clothing less often than on clothing already worn for a day.
Experiment
Container A
Container B
Container C
Container D
1
15
7
12
5
2
19
5
9
3
3
12
4
14
6
4
18
6
13
4
5
19
6
10
3
Justify a suitable conclusion for this investigation. (3 marks)
The normal Huntingtin protein has 10−26 repeats of CAG. In Huntington’s 2 disease there are 37−80 repeats.
Diagram 1 shows a pedigree of a family known to be affected by Huntington's disease. Diagram 2 shows the results of gel electrophoresis on fragments of DNA from chromosome four, known to be altered in Huntington's disease.
Diagram 1
Diagram 2
Predict whether individuals \(S\) and \(U\) will be affected by Huntington's disease, and if so, at what age. Use data from the diagrams to justify your answer. (3 marks)
--- 6 WORK AREA LINES (style=lined) ---
→ Individual \(U\) has the same number of CAG repeats as individual \(Q\) and therefore would be expected to be affected by Huntington’s at the same age, 45.
→ Individual \(S\) has a normal number of repeats (between 10-26) and much like individual \(P\) who has the same number of repeats, he is not expected to be affected by Huntington’s.
→ Individual \(U\) has the same number of CAG repeats as individual \(Q\) and therefore would be expected to be affected by Huntington’s at the same age, 45.
→ Individual \(S\) has a normal number of repeats (between 10-26) and much like individual \(P\) who has the same number of repeats, he is not expected to be affected by Huntington’s.
The normal Huntingtin protein has 10–26 repeats of CAG. In Huntington's disease there are 37–80 repeats. This leads to an alteration in the structure of the protein. The graph shows the relationship between the age of onset of Huntington's disease and the number of CAG repeats. Explain the relationship between the number of CAG repeats and the age of onset of Huntington's disease. (2 marks) --- 5 WORK AREA LINES (style=lined) --- → The graph shows that as the number of CAG repeats is increased, the age of the onset of Huntington’s decreases. → For example, someone that has 40 repeats will experience the onset of Huntington’s at 60 years old, but someone with 120 repeats will experience symptoms and/or exhibit signs shortly after their birth. → The graph shows that as the number of CAG repeats is increased, the age of the onset of Huntington’s decreases. → For example, someone that has 40 repeats will experience the onset of Huntington’s at 60 years old, but someone with 120 repeats will experience symptoms and/or exhibit signs shortly after their birth.
Huntington's disease is an autosomal dominant genetic disease. Using the pedigree, justify the genotype of individual \(H\). In your answer, refer to the letters on the pedigree to identify individuals. (3 marks) --- 6 WORK AREA LINES (style=lined) --- → Let \(S\) be the dominant allele for Huntington’s and \(s\) be the recessive allele. → As individual \(H\) is affected, she could genotype either \(SS\) or \(Ss\). → However individual \(H\) has children (\(J\) and \(L\)) that are not affected and thus have genotype \(ss\). This is only possible if she has the recessive allele, and therefore individual \(H\) must have genotype \(Ss\). → Let \(S\) be the dominant allele for Huntington’s and \(s\) be the recessive allele. → As individual \(H\) is affected, she could genotype either \(SS\) or \(Ss\). → However individual \(H\) has children (\(J\) and \(L\)) that are not affected and thus have genotype \(ss\). This is only possible if she has the recessive allele, and therefore individual \(H\) must have genotype \(Ss\).
--- 6 WORK AREA LINES (style=lined) --- --- 6 WORK AREA LINES (style=lined) --- a. → The front of the eye is made up of the cornea, lens and pupil. → These work together to direct light onto the retina at the back of the eye so that we can see. → The lens is controlled by ligaments which change the shape of the lens to allow the focus of light onto the retina from objects at varying distances. → If someone suffers from myopia (short-sightedness) their lens is too large or their ligaments are unable to bend the lens correctly, causing the focal point to fall short of the retina. b. → Glasses are a technology that help people with myopia. → Glasses with a concave lens will diverge light rays before they hit the eye lens, allowing the focal point to fall further back. → Lens technology fine tunes this effect so the focal point hits the retina optimally and perfect sight is restored. Answers could also include → A convex lens to address hyperopia (long-sightedness). → Laser surgery (LASIK) which uses a laser to flatten or steepen the cornea, addressing myopia, hyperopia and astigmatism. → Cataract surgery to replace a clouded/yellow lens. a. → The front of the eye is made up of the cornea, lens and pupil. → These work together to direct light onto the retina at the back of the eye so that we can see. → The lens is controlled by ligaments which change the shape of the lens to allow the focus of light onto the retina from objects at varying distances. → If someone suffers from myopia (short-sightedness) their lens is too large or their ligaments are unable to bend the lens correctly, causing the focal point to fall short of the retina. b. → Glasses are a technology that help people with myopia. → Glasses with a concave lens will diverge light rays before they hit the eye lens, allowing the focal point to fall further back. → Lens technology fine tunes this effect so the focal point hits the retina optimally and perfect sight is restored. Answers could also include → A convex lens to address hyperopia (long-sightedness). → Laser surgery (LASIK) which uses a laser to flatten or steepen the cornea, addressing myopia, hyperopia and astigmatism. → Cataract surgery to replace a clouded/yellow lens.
Consider the following nuclear reaction \({ }_{\ \ 6}^{12} \text{C} +{ }_1^1 \text{H} \rightarrow{ }_5^9 \text{B} +{ }_2^4 \text{He}\) The masses of the isotopes in this process are shown in the table. --- 6 WORK AREA LINES (style=lined) --- \(52.164\) \(\text{MeV}\) or \(8.357 \times 10^{-12}\) \(\text{J}\)
Isotope
Mass (\(u\))
\({ }_{\ \ 6}^{12} \text{C}\)
12.064
\({ }_5^9 \text{B}\)
9.013
\({ }_2^4 \text{He}\)
4.003
\({ }_1^1 \text{H}\)
1.008
\(\text{Mass defect}\)
\(=m_r-m_p\)
\(=(12.064+1.008)-(9.013+4.003)\)
\(=0.056\) \(\text{u}\)
\(\text{Energy released}\)
\(= 0.056 \times 931.5\) \(\text{MeV}\)
\(=52.164\) \(\text{MeV}\)
\(=8.357 \times 10^{-12} \) \(\text{J}\)
The diagram represents one type of electric motor. Describe the function of part \( X \). (2 marks) --- 5 WORK AREA LINES (style=lined) --- → Part \(X\) is a split ring commutator and its function is to reverse the direction of the current through the arms of the armature. → This ensures that unidirectional torque is maintained in the DC motor so that it continues to rotate in the same direction. → Part \(X\) is a split ring commutator and its function is to reverse the direction of the current through the arms of the armature. → This ensures that unidirectional torque is maintained in the DC motor so that it continues to rotate in the same direction.
An electron is travelling at 3.0 \(\times\) 10\(^{6}\) m s\(^{-1}\) in the path shown.
Calculate the magnetic field required to keep the electron in the path. (3 marks)
--- 6 WORK AREA LINES (style=lined) ---
\(1.7 \times 10^{-6}\) \(\text{T}\)
→ The force from the magnetic field on the electron provides the centripetal acceleration for it to travel in uniform circular motion.
| \( F_B\) | \(=F_c\) | |
| \(qvB\) | \(=\dfrac{mv^2}{r}\) | |
| \(B\) | \(=\dfrac{mv}{qr}\) | |
| \(=\dfrac{9.109 \times 10^{-31} \times 3.0 \times 10^6}{1.602 \times 10^{-19} \times 10}\) | ||
| \(=1.7 \times 10^{-6}\) \(\text{T}\) |
→ Magnetic field strength required = \(1.7 \times 10^{-6}\) \(\text{T}\).
The James Webb Space Telescope (JWST) is sensitive to wavelengths from 6.0 \(\times\) 10\(^{-7}\) m to 2.8 \(\times\) 10\(^{-5}\) m. What is the minimum photon energy that it can detect? (3 marks) --- 6 WORK AREA LINES (style=lined) --- \(E_{\text{min}}= 7.1 \times 10^{-21}\) \( \text{J}\) → The minimum photon energy corresponds to the minimum frequency. → Minimum frequency occurs at the maximum wavelength as frequency and wavelength are inversely proportional.
\(E_{\text{min}}\)
\(=hf\)
\(=\dfrac{hc}{ \lambda}\)
\(=\dfrac{6.626 \times 10^{-34} \times 3 \times 10^8}{2.8 \times 10^{-5}} \)
\(=7.1 \times 10^{-21}\) \(\text J\)
A spacecraft passes Earth at a speed of 0.9\(c\). The spacecraft emits a light pulse every 3.1 \(\times\) 10\(^{-9}\) s, as measured by the crew on the spacecraft. What is the time between the pulses, as measured by an observer on Earth? (3 marks) --- 6 WORK AREA LINES (style=lined) --- \(7.1 \times 10^{-9}\ \text{s} \)
\(t\)
\(=\dfrac{t_o}{\sqrt{(1- \frac{v^2}{c^2})}} \)
\(=\dfrac{3.1 \times 10^{-9}}{\sqrt{(1-\frac{(0.9c)^2}{c^2})}}\)
\( =\dfrac{3.1 \times 10{-9}}{\sqrt{(1-{0.9}^2)}} \)
\(=7.1 \times 10^{-9}\ \text{s} \)
A Hertzsprung–Russell diagram is shown. --- 4 WORK AREA LINES (style=lined) --- --- 6 WORK AREA LINES (style=lined) --- a. The luminosity of a star can be determined by its size and temperature. Other variables could include: → Mass, colour and the power output of a star. b. Differences: → Star A is a main sequence star and is therefore fusing hydrogen to helium in its core via both the proton-proton chain and CNO cycle whereas Star B is a white dwarf star and therefore has no fusion taking place in its core. → Star A has a greater luminosity compared to Star B. → Star A is a younger star then Star B which is at the end of its lifecycle. Other differences could include: → Mass → Radius or size a. The luminosity of a star can be determined by its size and temperature. Other variables could include: → Mass, colour and the power output of a star. b. Differences between stars: → Star A is a main sequence star and is therefore fusing hydrogen to helium in its core via both the proton-proton chain and CNO cycle whereas Star B is a white dwarf star and therefore has no fusion taking place in its core. → Star A has a greater luminosity compared to Star B. → Star A is a younger star then Star B which is at the end of its lifecycle. Other differences could include: → Mass → Radius or size
The chart shows part of a nuclear decay series beginning with uranium.
Which option correctly identifies \(X\) and \(Y\) and the process by which each was produced?
| \(X\) | \(Y\) | |
| A. |
\({ }_{\ \ 90}^{234}\text{Th}\) alpha decay |
\({ }_{\ \ 91}^{234}\text{Pa}\) beta decay |
| B. |
\({ }_{\ \ 90}^{234}\text{Th}\) alpha decay |
\({ }_{\ \ 91}^{234}\text{Pa}\) alpha decay |
| C. |
\({ }_{\ \ 91}^{234}\text{Pa}\) beta decay |
\({ }_{\ \ 90}^{234}\text{Th}\) beta decay |
| D. |
\({ }_{\ \ 91}^{234}\text{Pa}\) beta decay |
\({ }_{\ \ 90}^{234}\text{Th}\) alpha decay |
\(A\)
→ During the decay process producing \(X\), the Atomic number decreases by 2 to 90 and the Mass number decreases by 4 to 234
→ Alpha decay producing \({ }_{\ \ 90}^{234}\text{Th}\)
→ During the decay process producing \(Y\), the Atomic number increases by 1 to 91 and the Mass number does not change.
→ Beta decay producing \({ }_{\ \ 91}^{234}\text{Pa}\)
\(\Rightarrow A\)
The graph shows the relationship between radiation intensity and wavelength for a black body at 4500 K.
Which statement describes the expected difference in the graph for a black body at 4000 K?
\(A\)
→ The total power output of a black body diminishes with decreasing temperature, resulting in lower intensity across all wavelengths for the 4000 K curve.
\(\Rightarrow A\)
A proton and a neutron travel at the same speed.
Which statement correctly explains the difference between their de Broglie wavelengths?
\(C\)
→ de Broglie wavelengths are given by the equation \( \lambda = \dfrac{h}{mv}\)
→ \( \lambda \propto \dfrac{1}{m} \)
∴ Since the mass of a neutron is slightly greater than the mass of a proton, the neutron will have a shorter wavelength.
\(\Rightarrow C\)
An electron would produce an electromagnetic wave when it is
\(D\)
→ Maxwell predicted that an accelerating charge will produce an electromagnetic wave.
→ As the electron moves at a constant speed in a circular path it must be experiencing an acceleration.
\(\Rightarrow D\)